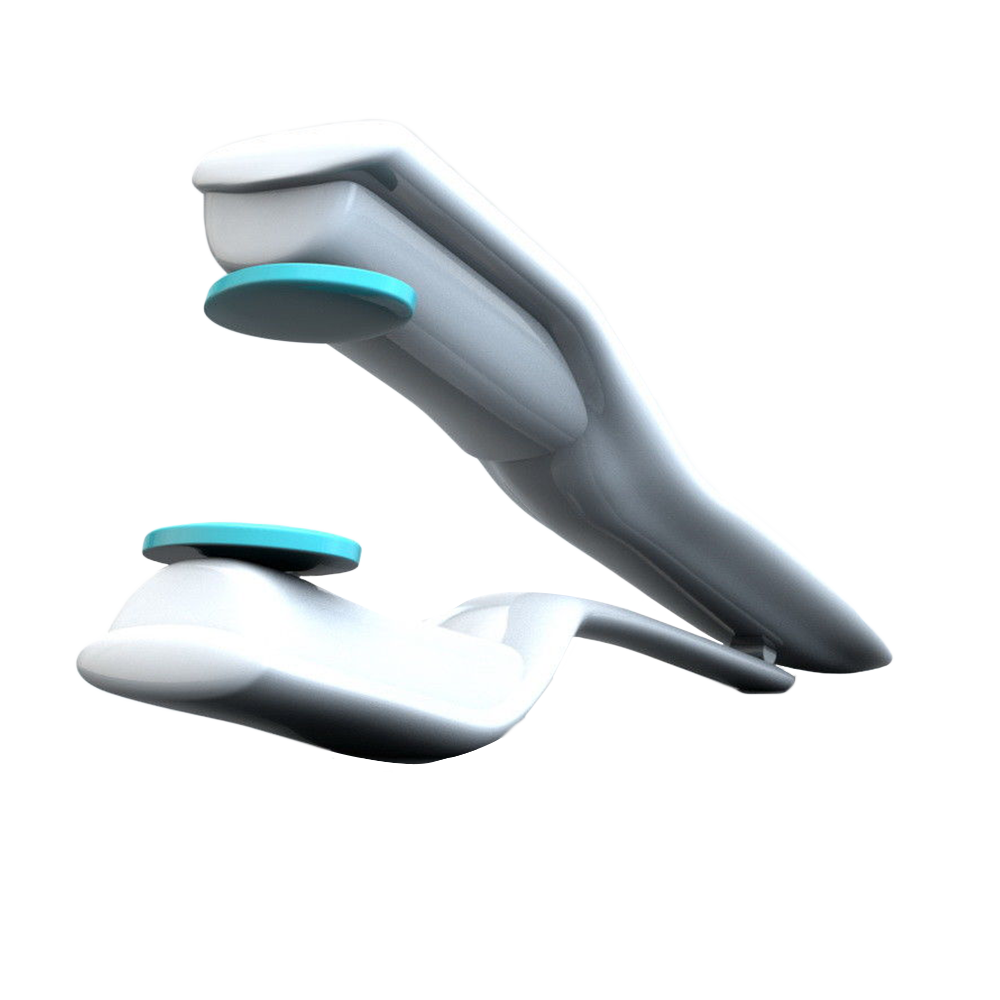
Description
Viberect X3 helps men overcome many forms of sexual dysfunction, including difficulty obtaining and maintaining an erection, rigidity issues, erectile dysfunction occurring after prostate surgery, and ejaculatory and orgasmic dysfunction. It is the only vibratory medical device cleared by U.S. FDA and the European Union for the treatment of ED.
Viberect Series X3 advanced penile vibratory system was developed by the highly acclaimed urologist, Dr. Kambiz Tajkarimi. The sonic vibration technology used in Viberect devices is patented and is proven to successfully trigger erections in men with (moderate-to-severe) erectile dysfunction and conditions related to post prostate cancer surgery, aging, multiple sclerosis, diabetes, hypertension (high blood pressure), and spinal cord injuries.
Viberect X3 System Includes:
- Viberect dual Mabuchi motor stimulation system
- One pair of medical soft pads (replaceable)
- User Manual and instructions
- Deluxe packaging with soft foam insert for storage
- Rechargeable batteries
- Low Profile shipping and packaging
- 1 Year Guarantee
Technical X3 Specifications:
- Made in The USA
- Vibration frequency: 70-110 Hertz Amplitude
- Vibration amplitude of stimulation: 3 mm
- Ni-Alu-Hydride Rechargeable batteries
- Voltage: 110-240 Volts
- US FDA, CE, Health Canada, ARTG
Certification: CE, US FDA, Health Canada, ARTG
FDA Statement: Compliant with medical device industry standards for electronic and plastic safety including ISO 10993-2, ISO 17025, ISO 13485-2003, CMDCAS, and EN 60601-3.
Precautions: Viberect use by men with spinal cord injuries carries the risk of Autonomic Dysreflexia. If you suffer from a spinal cord injury, please seek advice from your health care provider.